
MEL Chemistry is a subscription box for kids ages 9-14 that sends all of the materials you need to complete 2-3 chemistry experiments every month. This is perfect for kids who are interested in science or for homeschool students!
This box was sent to us at no cost for review. (Check out the review process post to learn more about how we review boxes.)

About MEL Chemistry
The Subscription Box: MEL Chemistry
The Cost: $34.90 per month
The Products: Everything you need to conduct 2-3 chemistry experiments each month safely in your own home!
Ships to: The U.S. and UK for free. International shipping to select countries is also available, although there may be additional shipping charges depending on location.
MEL Chemistry "Copper" Review June 2020
This month our experiments' theme is "Copper" and the contents were listed on the back of our box. You may note that they list a few items we need to get from our Starter Kit.
Note: The following items did not come in this month's kit!
Starter Kit
Your free starter kit (valued at $50) will come in your first shipment. It includes all of the equipment you'll need to conduct your experiments each month and even includes some accessories for your tablet or smartphone to help you access all of the information they provide in their apps. To see more details about everything we received in our kit, you can check out our review here!
VR Headset
Also included with our Starter Kit was this virtual reality headset. This cardboard headset folds up easily and then you slip your phone into the slot and use their free MEL VR app.
The VR app opens up in a lab where we have the opportunity to learn about different molecules and atoms and explore them closely. This is so great for gaining a deeper understanding of what happens throughout our experiments and as a fun extension to our lessons!
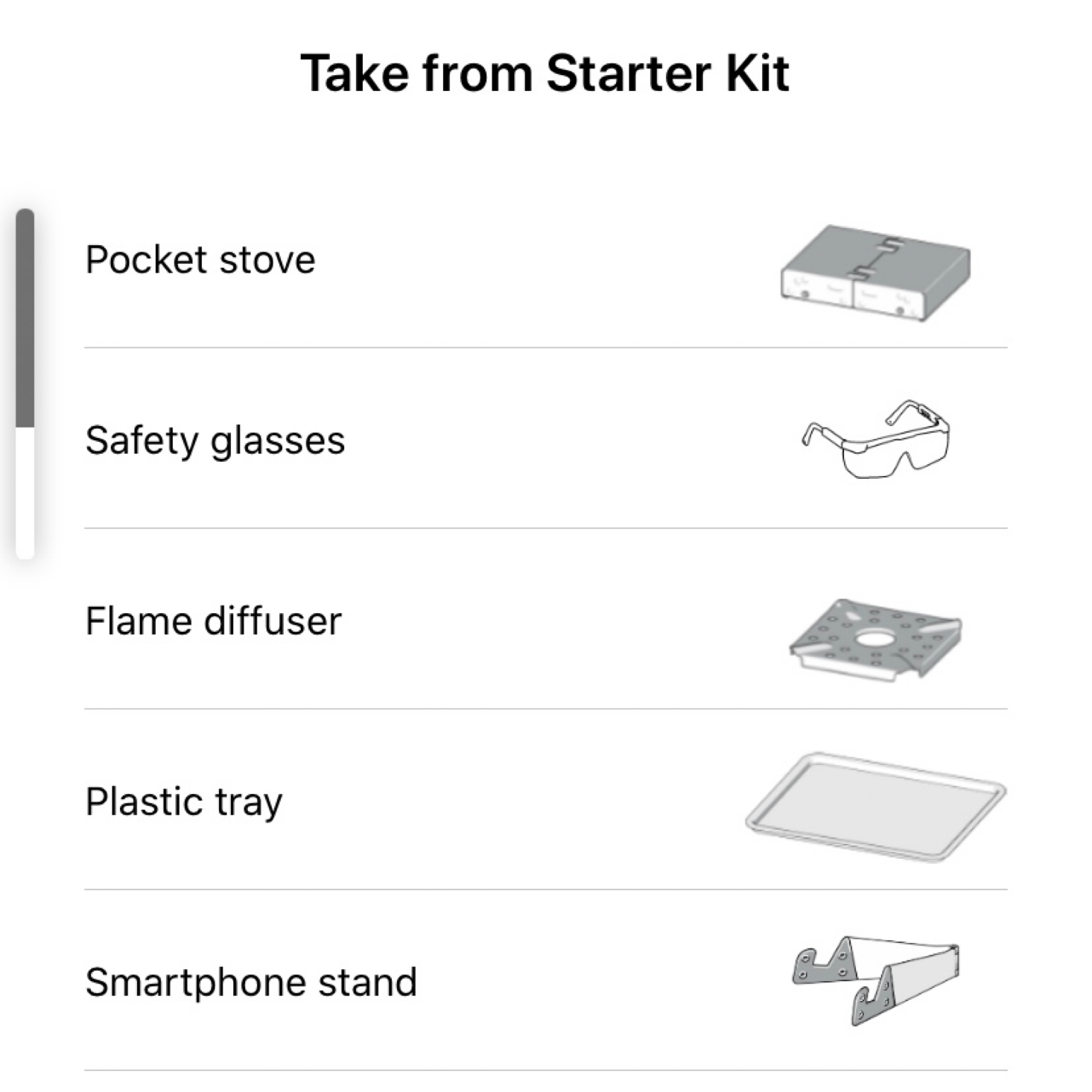
The first three items are those that we need each month: a tray to work on, our safety goggles, and our phone/tablet stand.
This month we needed quite a few things from our kit such as our beakers and flask, our funnel, and our stove.
Inside our box was an instruction sheet for the parents including some safety warnings, advice, and general first aid information. The directions for the projects are pretty thorough and can be found in this booklet, on the individual experiment cards or in the app. Having this separate sheet for the parents is great so kids can work independently, but parents can also follow along for supervising.

The projects come neatly organized with all of our reagents neatly stored in plastic bags with absorbers in each to help in case of any spills. Let's get to our first experiment!
Experiment 1: Why So Blue?
Our first experiment was titled "Why So Blue?" For each experiment, they provide a card like this one that contains all of the necessary information such as safety, disposal, and the directions complete with colored illustrations. The front gives a brief descriptor of what to expect and also provides a scale to rate the difficulty and danger of the activity as well as the duration. On the back is a little box we can scan with our app which allows us to open directly to the experiment on our phone or tablet. Of course, this is optional since you have everything you need on the experiment card, but I really like the interactiveness of the app instructions.
They included this card with a QR code for easy access to the app.
I set the phone in our stand and we clicked the "start experiment" button to begin. They provided information on the expected results, troubleshooting, and even a scientific description of what happens during the experiment. There is also an easy way to set a reminder to conduct your experiment!
There is also a safety section that we always check out first. Then we get prepared by putting on our gloves and our protective eyewear. They sent a pair of small gloves and a pair of large gloves so that an adult assistant can help.
The app provides thorough, detailed instructions. Some of the instructions are even animated which is pretty cool and usually if it is a timed experiment, they will have a little built-in timer on that page for you to use. They also have little "help" tips at the bottom in case you get confused about what to do.
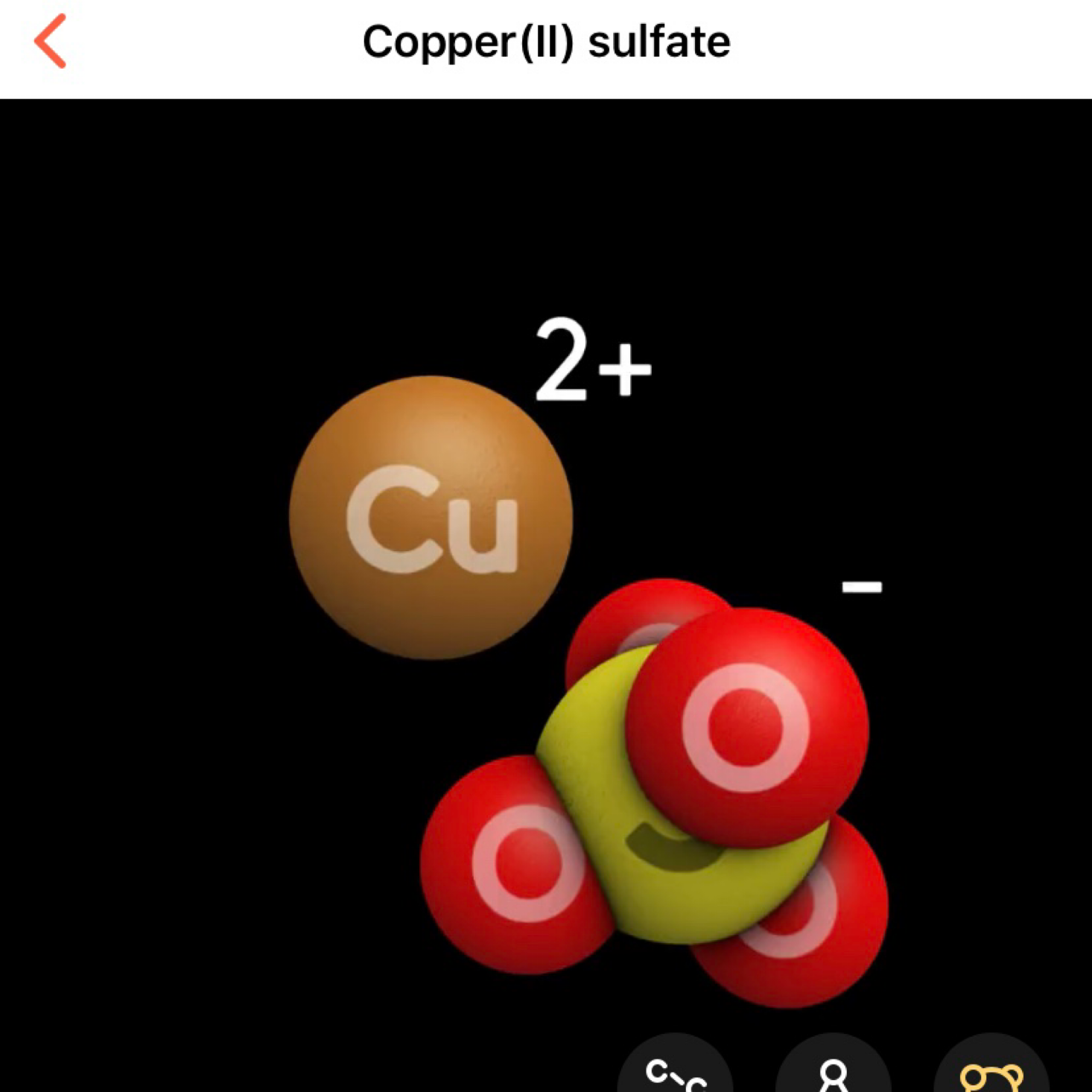
All of our reagents are clearly labeled and include information about any hazards. They provided enough materials for us to complete our experiments several times. Each label also contains a little code that we can scan with our forward-facing camera to open up for more information about our chemicals. We are able to view it in 3D or even in virtual reality by using the VR app and our headset. For this experiment, we are using Copper Sulfate Pentahydrate.
The app gives so much detail about each of the reagents including a description, hazards, precautions, and even a link to the Wikipedia page for further information. So thorough! And it's even more fun if you interact through the VR app!
They always provide a little paper clip for us to use to open our bottles which have tiny rubber stoppers in them so they don't leak during transportation along with some of these little measuring spoons. We also received some thermo-stickers to place on the equipment that would get heated.
My assistants got prepared with their gloves and safety goggles.
I set up our stove and lit the tealight candle that they included for us.
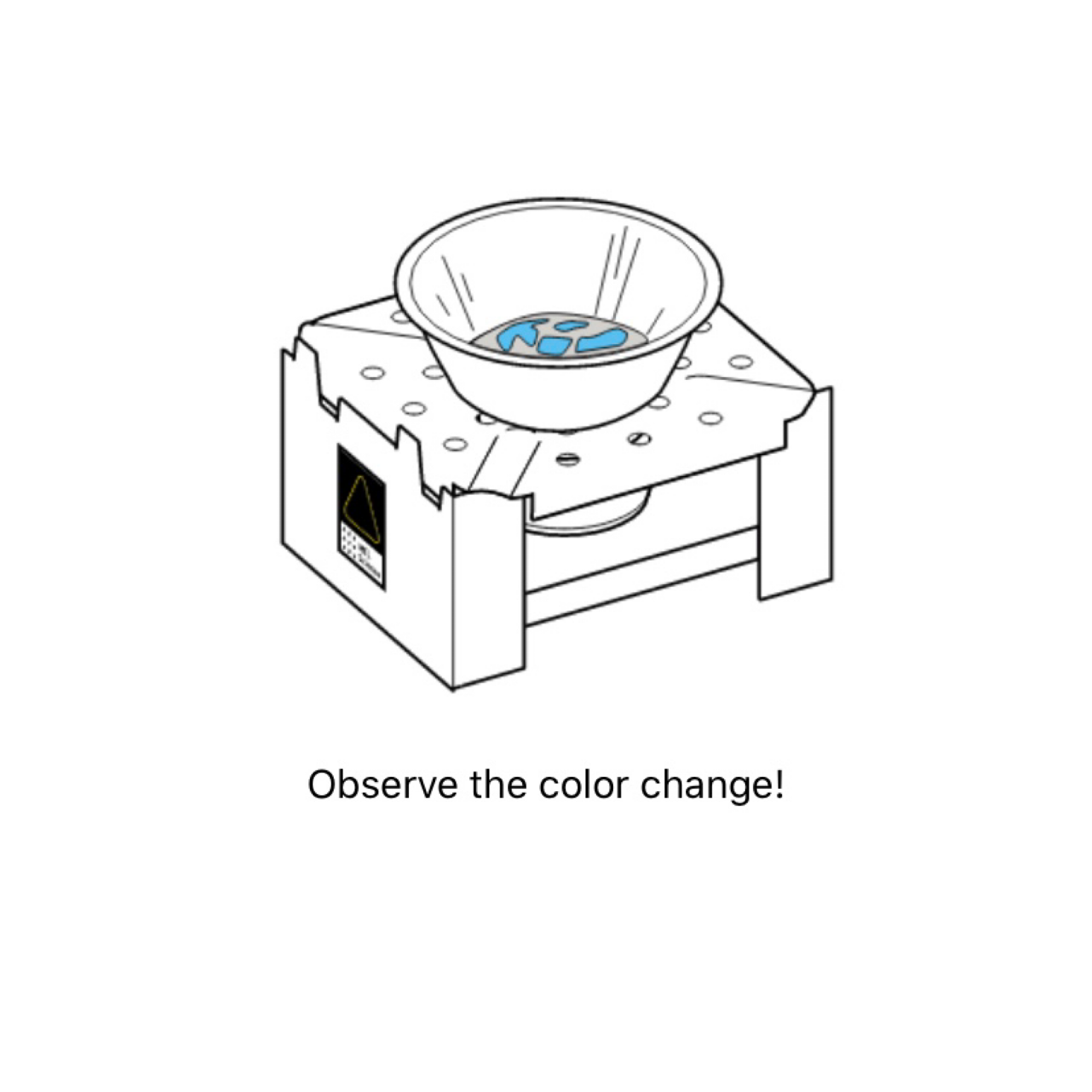
My daughter then measured out a large spoonful of the copper sulfate and placed it in one of the little foil pans. Then I placed it on top of the stove and we observed.
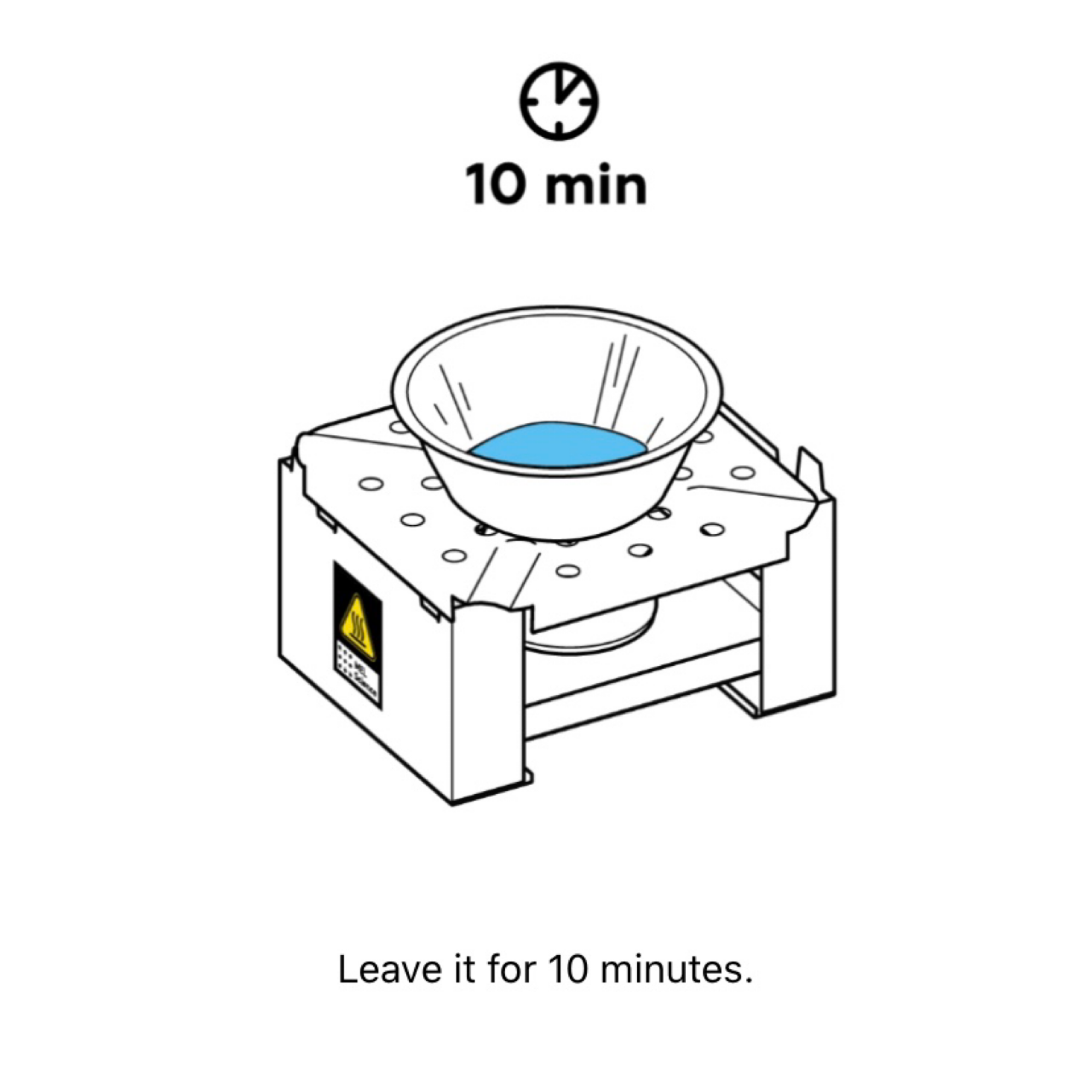
As we observed, the crystals began to turn from blue to white. We had quite a long wait for them to turn completely white, but we kept moving and did our second experiment while we waited.
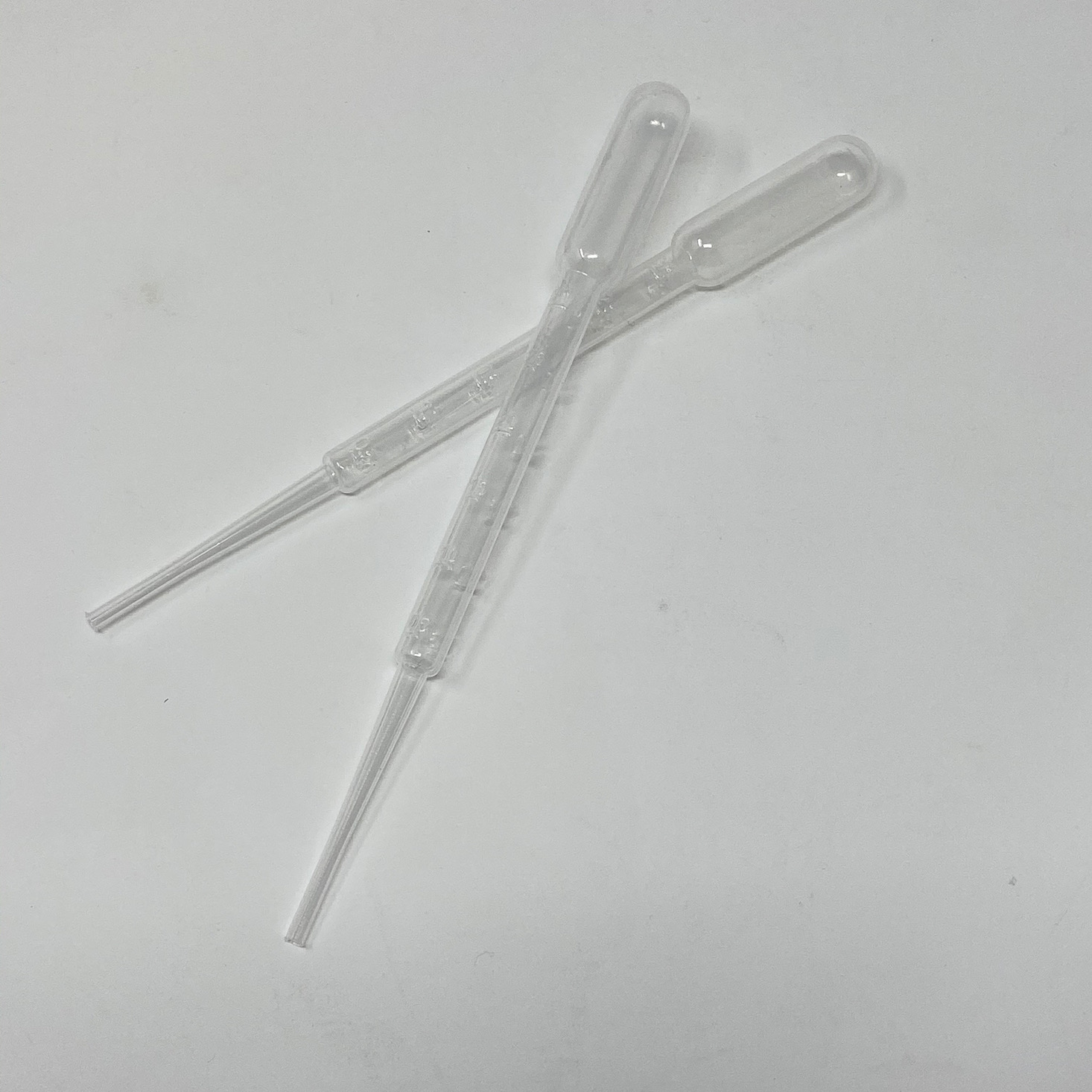
Once 15 minutes had passed, we blew out the flame and I got a little beaker of water and grabbed one of the droppers so the kids could add some water drops to it and observe the results.
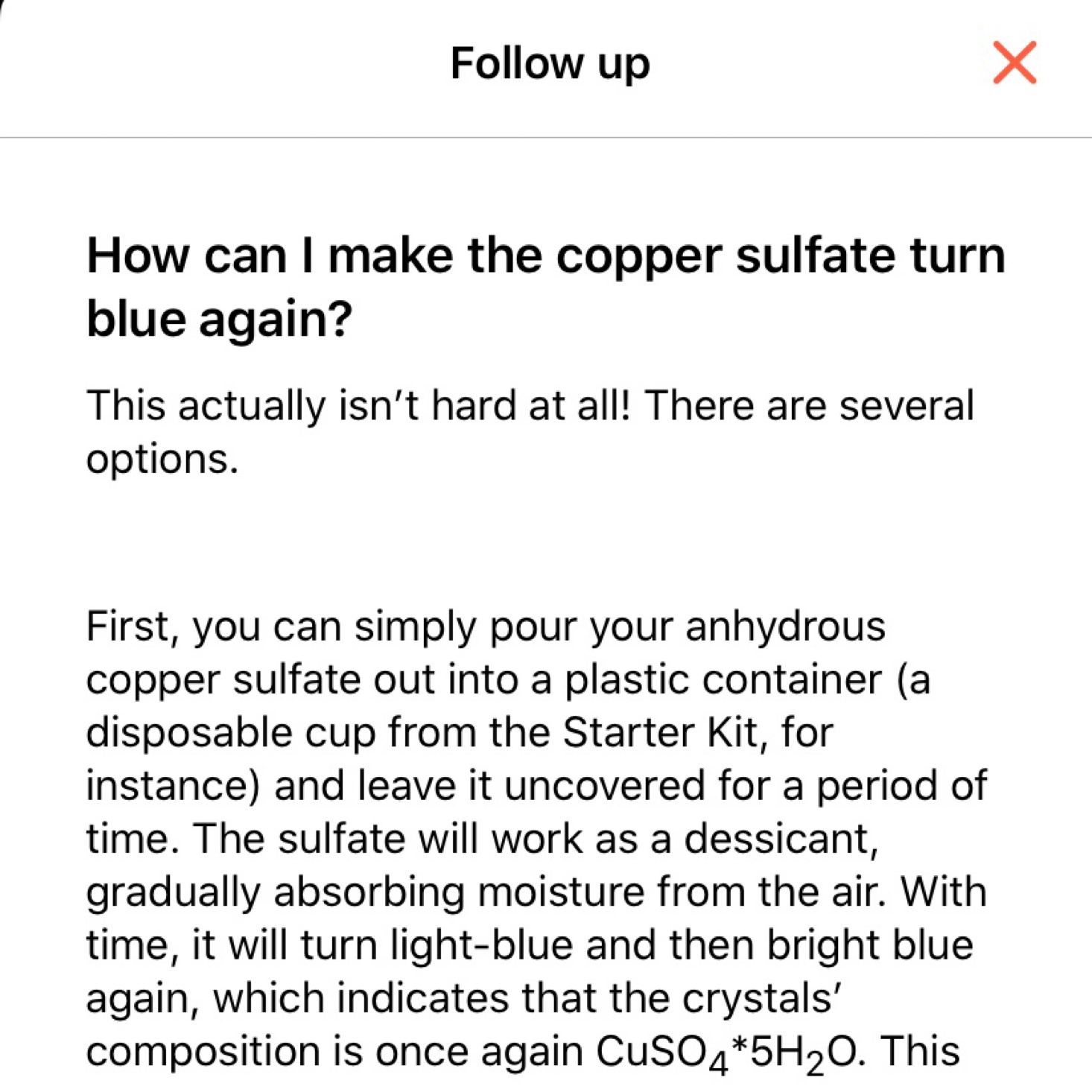
They were delighted to find that it turned back to blue after adding water and we were able to talk about what was happening and why thanks to all of the educational information in the app and the instructions card.
At the end of our experiments, our app asks us to rate the experiment and then there is a section on how it works which includes helpful information such as a scientific description, interesting facts, and disposal instructions, all of which we had access to from the main page of this experiment as well.
Experiment 2: Copper Complexes
The next experiment was "Copper Complexes" and we used several different reagents including liquid copper sulfate, sodium chloride, disodium disulfate, and ammonium carbonate.
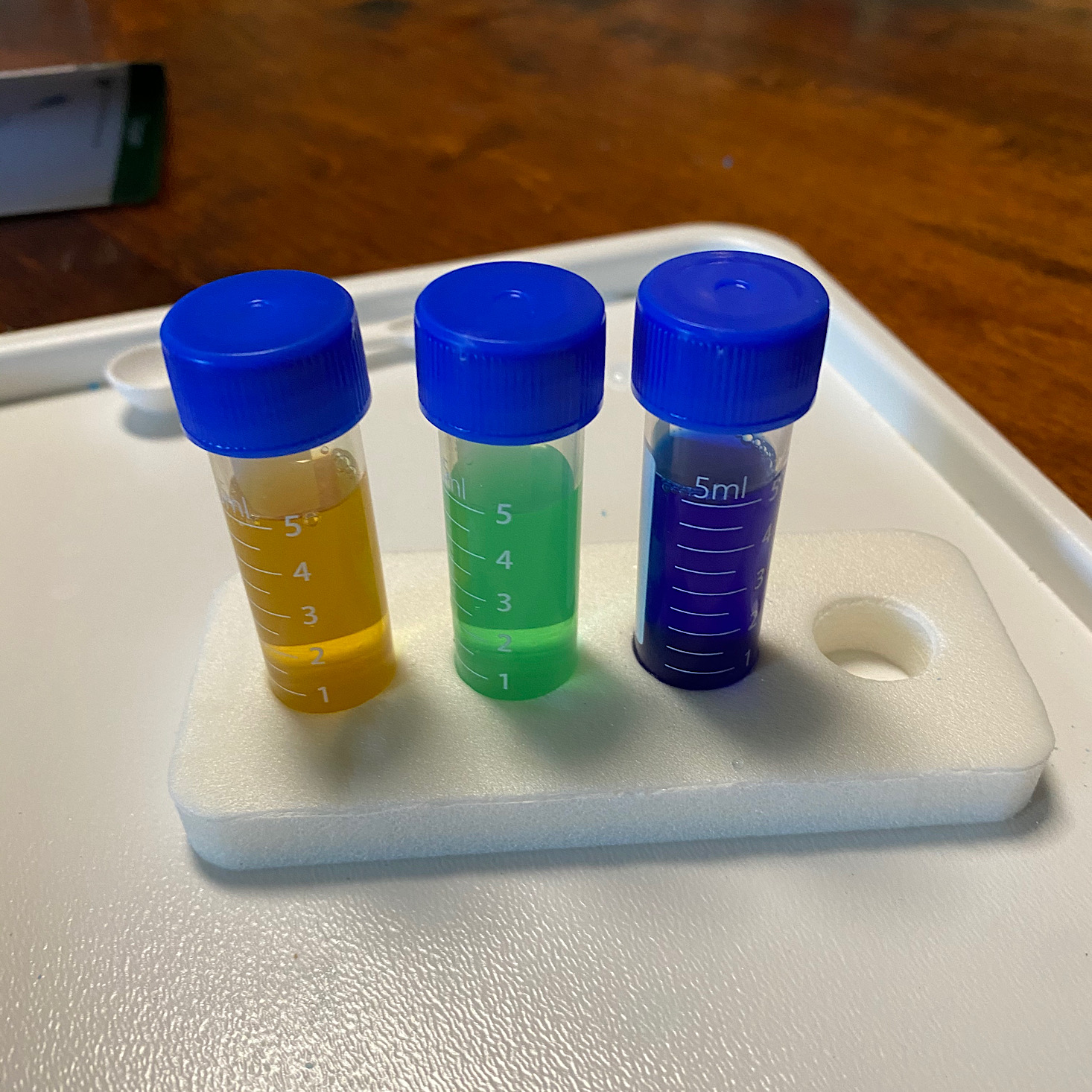
They provided this foam stand and these tiny plastic vials for us to use. I got my daughter set up with three of them with their lids off.
Then she began to fill them according to the directions. Each got 4 mL of the reagents and then we topped all three of them off with 1 mL of the copper sulfate.
Then we replaced the lids and my daughter shook them all up.
She thought it was pretty cool how they all changed colors! The reactions were almost immediate, but much easier to see once she shook them all up.
Experiment 3: Copper Citrate
The last experiment was "Copper Citrate" and it was much more involved!
In addition to the sodium citrate and copper sulfate pentahydrate, they also provided some stirring sticks, two filters which were made to fit right into our funnel, and a few splints for our last step.
First, I had my son apply the thermosticker to our glass beaker. Then my daughter used one of the plastic vials from our second experiment and measured out 5 mL of the copper sulfate pentahydrate crystals. My son then dumped those into the beaker.
Then I had my daughter stir while I added some boiling water to it. We stirred for 10 seconds.
Then my son added one bottle of sodium citrate to it and stirred for another 30 seconds.
We let the beaker sit undisturbed for 15 minutes and prepared our flask by adding the funnel and filter on top of it.
When the 15 minutes were up, my daughter carefully and slowly poured the mixture into the filter. It took a long time to filter through (so long that I lost my helpers!) and once I got to the last little bit, I used the wooden stick to scrape out the sediment at the bottom. Then I waited for it all to filter through.
Once that filtered through, I added some more water to the filter and then it was time to set it aside for a full 24 hours before our next step.
The next day, I used a measuring spoon to transfer all of the precipitate to a foil pan. Then using the back of the spoon, I crushed it up.
Next, I prepared a beaker full of boiling water and we set the foil pan on top of it. We let it sit and began to observe what happened. It was pretty cool to see the colors change!
Then it was time to light one of our splints on fire and we touched it to the crystals at different spots. You can see they started to burn and little embers were sparking throughout the pan.

The heat kept spreading through our pan until almost all of the crystals were brown! This was a fun one to watch since it changed so much. The little sparks were really cool to see and since they were so small they never left the pan, so I felt it was very safe, too!
Verdict: This month's MEL Chemistry lessons felt like some pretty substantial experiments! Since one experiment included 24 hours of wait time, I think this could easily be a 2-day homeschool chemistry lesson! There was so much to learn and observe and there was a great deal of measuring and waiting, which I think are important lessons that can sometimes be overlooked. Overall, this was another thorough and educational look at Chemistry that we enjoyed!
To Wrap Up:
Can you still get this box if you sign up today? Yes, it is possible that you will receive this set. From MEL Chemistry:
"The topics are looped such that the start date of your subscription does not matter; you will eventually receive all the experiment sets."
Check out all of our MEL Chemistry reviews and our list of the best subscription boxes for kids, as recommended by MSA readers!
Keep Track of Your Subscriptions: Add MEL Chemistry to your subscription list or wishlist!
What do you think of MEL Chemistry?



























































































































Please do not enter your email address in the Name field or in the comment content. Your email address will not be published. Required fields are marked *. Remember to post with kindness and respect. Comments with offensive language, cruelness to others, etc will not be approved. See our full comment policy here.